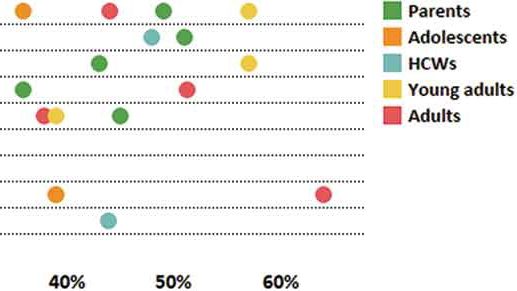
Overview
The Vaccine Confidence Project™ is a WHO Centre of Excellence on addressing Vaccine Hesitancy, and is engaged in many strands of research pertaining to vaccine hesitancy, including (but not limited to) the projects described below. VCP Founder and Co-Director Professor Heidi. J Larson also served on the WHO SAGE Working Group on vaccine hesitancy.
ADVANCE
ADVANCE (Accelerated development of vaccine benefit-risk collaboration in Europe) was a five-year IMI-funded project which started in October 2013. It was a unique project which aimed to review, develop and test methods, data sources and procedures which should feed into a blueprint of an efficient and sustainable pan-European framework that could rapidly deliver robust quantitative data for the assessment of the benefits and risks of vaccines that are on the market. Such a framework would allow regulators and public health authorities to make fast, informed decisions regarding vaccination strategies, and help to maintain public confidence in vaccines, particularly when questions are raised in the media about the safety of specific vaccines.
The Vaccine Confidence Project™ was responsible for analysing public concerns and perceptions related to benefits and risks of vaccines, analysing key issues and gaps about perception and knowledge on benefits and risks of vaccines, and developing a strategy for public communication in the context of vaccine benefit-risk monitoring.
More information about the project can be found here: https://www.imi.europa.eu/projects-results/project-factsheets/advance
Vaccine Hesitancy in Europe
The Vaccine Confidence Project™ has been involved in various projects with the European Centre for Disease Prevention and Control on understanding vaccine hesitancy in Europe, particularly among healthcare workers. The latest project, started in September 2016 and aimed to better understand HPV vaccine hesitancy in Europe.